mFluor™ Blue 615 SE
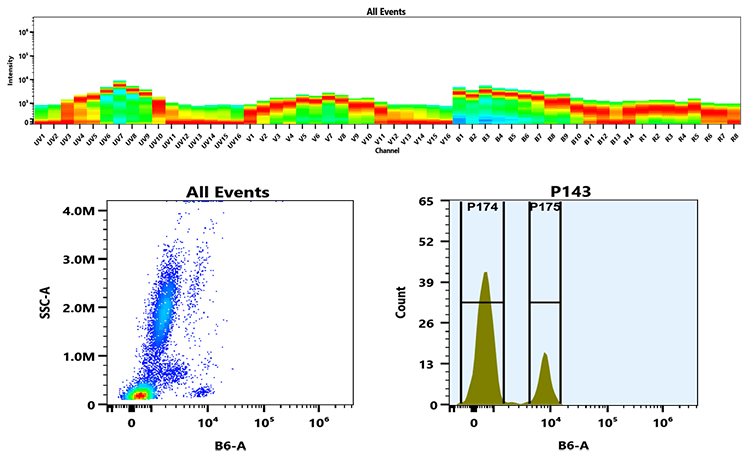
mFluor™ Blue 615 dye can be well excited with blue laser at 488 nm. It has a huge Stokes shift with emission ~615 nm. mFluor™ Blue 615 dyes are water-soluble, and the protein conjugates prepared with mFluor™ Blue 615 dyes are well excited at 488 nm to give red fluorescence. mFluor™ Blue 615 dye and conjugates are excellent blue laser reagents for flow cytometry detections. Compared to RPE, mFluor™ Blue 615 dyes are much more photostable, making them readily available for fluorescence imaging applications while it is very difficult to use RPE conjugates for fluorescence imaging applications due to the rapid photobleaching of RPE conjugates. It is also a unique fluorochrome for spectral flow cytometry since there are very few existing dyes that have this spectral profile. In addition, mFluor™ Blue 615 dyes are small organic dyes that have much smaller size than the macromolecular RPE, thus potentially less affinity interference for the molecules to be labelled.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 1162 | 1 mg | Price |
Physical properties
| Molecular weight | 815.87 |
| Solvent | DMSO |
Spectral properties
| Extinction coefficient (cm -1 M -1) | 40000 1 |
| Excitation (nm) | 510 |
| Emission (nm) | 615 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on March 23, 2026

